 |
 |
 |
 |
GILLESPIE BORATE: : 50 pound Bag
Gerstley Borate SubstituteGerstley Borate is no longer available.
Use Gillespie Borate as a substitute.
Gerstley Borate was a highly variable material and could be different from batch to batch. Gillespie borate is manufactured to meet the most typical analysis of the Gerstley borate so you should find this to be a perfect substitute and more dependable.
Please test your formulas before using on your work.
Gillespie Borate Fact Sheet
Description : Gillespie Borate is a blended borate mineral for use in glaze formulas replacing Gerstley Borate on a pound for pound basis.
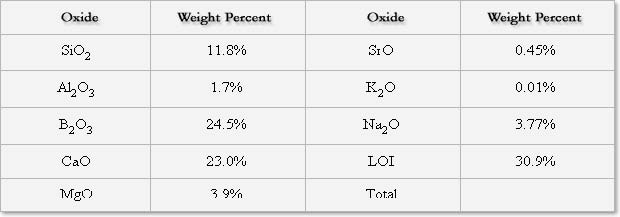
Chemical Analysis [Typical]
Features
Consistency -- Manufactured from high purity refined raw materials, Gillespie Borate is highly consistent from batch to batch.
Oxide Match -- Virtually identical to Gerstley Borate, this product requires no adjustment to formula and no extra additives are needed.
Mineral Match -- Gillespie Borate provides the same minerals to the glaze as Gerstley Borate, thus imparting the same glaze activity, texture and effects.
Particle Size Match -- As a -200 mesh material, Gillespie Borate combines well with other glaze raw materials.
Plasticity Match -- A high colloid and clay content gives the plasticity necessary for many applications.
Reduced Glaze Defects -- Our laboratory studies indicate that Gillespie Borate gives increased glaze surface smoothness and a reduction in crawling and pinhole defects.
Lower In Off-Color Impurities -- As a refined blended material, Gillespie Borate contains few impurities and produces brighter glazes and colors.
Applications
Raku, Earthenware, Stoneware and Porcelain -- Excellent over a wide temperature range from below cone 06 to cone 10
Special Effects -- Excellent glaze texture and activity for specialty glazes.
Colored Glazes -- GB works well with most pigments and has an exceptionally wide color palette.
Finding a replacement for Gerstley Borate
Gerstley borate is a complex ore but a major benefit of the unrefined naturally formed combination of minerals is its ability to produce variegated surfaces in glazes. Potters firing electric kilns which have oxidation atmospheres often produce flat static glaze effects much like bathroom tile colors. Carbon based fueled kilns, such as natural gas, propane or wood have the capacity to fire in oxidation, neutral, and reduction atmospheres which can introduce varied glaze colors and surfaces. If it wasnt for this unique characteristic of Gerstley borate it would have been abandoned when the first series of its faults appeared.
Gerstley borates wide swings in chemical composition are a factor in finding a direct substitute. Its like trying to hit a moving target when deciding on what chemical formula to use when developing a substitute.
However, Gillespie borate, a product produced by Hammill & Gillespie of New Jersey, has proven to be a reliable one-for-one substitute. In many instances it is difficult to arrive at such a close match as many raw materials are not mineralogically pure when removed from the earth. Any potential substitute has to match on many points and produce the same results in fired glazes.
Gerstley Borate has been a valuable glaze constituent for many years and is a key part of many interesting and successful glazes over a range of firing conditions. As the supply has dwindled Hammill & Gillespie's technical staff went to the lab and began experimenting. Beginning with x-ray diffraction to determine the mineral constitution of Gerstley borate and following up with other physical and chemical tests, they fully characterized the material and identified three key characteristics: extended particle size distribution including a colloid fraction, a specific and unique distribution of oxides, and a fired property that promotes a variegated color in many glaze compositions.
To generate a replacement, a new material was created: Gillespie borate, which is virtually identical to Gerstley borate in terms of physical, mineralogical and chemical composition. Gillespie borate is comprised principally of ulexite (sodium calcium borate), with small amounts of colemanite (calcium borate) and other minerals -- just like Gerstley borate. From a pre-fired perspective the most critical element in the new material is its colloid content. Containing approximately 10% clay-like materials, Gillespie borate disperses well in water and makes a reasonably stable glaze slip in the neat state. Further clay addition at the point of use is required to further improve the stability of the slip and pre-fired properties such as strength and adherence. In the fired state, Gillespie borate is virtually identical to Gerstley borate by virtue of the chemical match of oxide content.
The special behavior of Gerstley and Gillespie borates that produces the unusual glaze effects results from two mechanisms: immiscibility and solubility. Boric oxide (B2O3), when provided to a glass as a coarse raw mineral (e.g. ulexite), is slightly immiscible with silicate glass matrices, and the differential surface energy of the molten phase drives a mass transfer process along the interface (Gibbs-Marangoni effect) that generates the characteristic variegated texture. The slight solubility of these borates also contributes to the mottled glaze surface via migration of soluble oxides, although this effect becomes exaggerated when mixed glazes are stored for long periods.
While Gillespie Borate has the beneficial fluxing and glaze development properties of Gerstley borate, it does not have the variability or impurity levels that characterized Gerstley borate. Gillespie borate is a precise mixture of pure natural minerals and is manufactured under close quality control supervision. Hence, Gillespie borate does not vary over time and is a consistent, reliable raw material. Since Gillespie borate does not contain the impurity levels that provided off-color to Gerstley borate glazes, you may find your glazes whiter and brighter. 2.
Recommendations for using Gillespie borate Do not use excess water in mixing the glaze.
Store the wet glaze in an airtight container.
When using any substitute material always test it in a small batch of glaze.
Test the glaze on the same clay body you will use in production.
After applying the glaze to several vertical test tiles place them throughout the kiln.
Footnotes
The exact chemical composition of Gillespie borate is proprietary to Hammill & Gillespie. Gillespie borate is a mixture of ulexite (Na2O.2CaO.5B2O3.16H2O) with various clay minerals (aluminum silicates), alkaline earth carbonates and silicates. This material contains approximately 25% B2O3, 23% CaO, 4% total alkali and 31% loss on ignition.

|
|
 |
 |
 |
 |

|




